Sayana Press
PATH
Solution Overview & Benefits
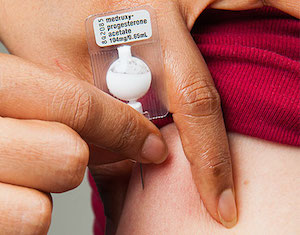
Injectable contraceptives are among the world’s most popular methods for preventing pregnancy, offering women safe and effective protection, convenience, and privacy. Sayana® Press, a new formulation and presentation of Depo-Provera®, offers the potential to improve contraceptive access for women worldwide.
Sayana Press is a three-month, progestin-only injectable contraceptive product packaged in the Uniject™ injection system, a small, prefilled, autodisposable device. It contains 104 mg of depot medroxyprogesterone acetate (DMPA) and is administered via subcutaneous injection.
History & Development
As a result of the Summit on Family Planning commitment, PATH and partners are supporting pilot introduction of Sayana Press in in Bangladesh, Burkina Faso, Niger, Senegal, and Uganda. Country introduction activities are expected to begin in the first quarter of 2014. Plans are under way to make Sayana Press available through normal delivery channels in the public, NGO and commercial sectors. The project’s evaluation component will generate information that builds upon the evidence base for decision-making about whether and how countries can include Sayana Press in family planning programs in the future. The pilot introduction units of Sayana Press are being procured by donors involved in the Summit on Family Planning.
This pilot introduction will complement small-scale Sayana Press acceptability studies and operational assessments completed in Senegal and Uganda in 2013. These studies generated information concerning the acceptability of Sayana Press among clients and a range of health care providers, including community-based workers.
Availability
Publicly available: yes
Countries where available: Worldwide
Price range (USD): Contact supplier for quote
Additional Information
Updates
If you are aware of any updates to the Sayana Press project please complete the form or send an email to [email protected]
Need help finding solutions that respond to your unique development challenges? Apply through our Project Accelerator service or Contact us directly for needs-based project consulting.